Neisseria gonorrhoeae
| Neisseria gonorrhoeae | |
|---|---|

| |
| Gram-stain of gonococcal urethritis. Note distribution in neutrophils and presence of both intracellular and extracellular bacteria. (CDC) | |
| Scientific classification | |
| Domain: | Bacteria |
| Phylum: | Pseudomonadota |
| Class: | Betaproteobacteria |
| Order: | Neisseriales |
| Family: | Neisseriaceae |
| Genus: | Neisseria |
| Species: | N. gonorrhoeae
|
| Binomial name | |
| Neisseria gonorrhoeae | |
| Synonyms | |
Neisseria gonorrhoeae, also known as gonococcus (singular) or gonococci (plural), is a species of Gram-negative diplococci bacteria first isolated by Albert Neisser in 1879.[3] As an obligate human pathogen, it primarily colonizes the mucosal lining of the urogenital tract; however, it is also capable of adhering to the mucosa of the nose, pharynx and rectum, and the eyes.[4] It causes the sexually transmitted genitourinary infection gonorrhea[5] as well as other forms of gonococcal disease including disseminated gonococcemia, septic arthritis, and gonococcal ophthalmia neonatorum.
It is oxidase positive and aerobic, and it survives phagocytosis and grows inside neutrophils.[5] Culturing it requires carbon dioxide supplementation and enriched agar (chocolate agar) with various antibiotics (Thayer–Martin). It exhibits antigenic variation through genetic recombination of its pili and surface proteins that interact with the immune system.[3]
Sexual transmission is through vaginal, anal, or oral sex.[6] Sexual transmission may be prevented through the use of barrier protection.[7] Perinatal transmission may occur during childbirth, and may be prevented by antibiotic treatment of the mother before birth and the application of antibiotic eye gel on the eyes of the newborn.[7] After an episode of gonococcal infection, infected persons do not develop immunity to future infections. Reinfection is possible due to N. gonorrhoeae's ability to evade the immune system by varying its surface proteins.[8]
N. gonorrhoeae can cause infection of the genitals, throat, and eyes.[9] Asymptomatic infection is common in males and females.[7][10] Untreated infection may spread to the rest of the body (disseminated gonorrhea infection), especially the joints (septic arthritis). Untreated infection in women may cause pelvic inflammatory disease and possible infertility due to the resulting scarring.[9] Diagnosis is through culture, Gram stain, or nucleic acid tests, such as polymerase chain reaction, of a urine sample, urethral swab, or cervical swab.[11][12] Chlamydia co-testing and testing for other STIs is recommended due to high rates of co-infection.[13]
Antibiotic resistance in N. gonorrhoeae is a growing public health concern, especially given its propensity to develop resistance easily.[14] This ability of N. gonorrhoeae to rapidly adapt to novel antimicrobial treatments has been seen several times since the 1930s with the introduction of sulfonamide treatments all the way to modern strains beginning to exhibit resistance to current ceftriaxone treatments.[15]
Microbiology
[edit]
Neisseria species are fastidious, Gram-negative cocci that require nutrient supplementation to grow in laboratory cultures. They are facultatively intracellular and typically appear in pairs (diplococci), resembling the shape of coffee beans. Members of this genus are non-spore-forming, capable of moving using twitching motility, and obligate aerobes (requires oxygen to grow). Of the 11 species which colonize humans, only two are pathogenic: N. gonorrhoeae and N. meningitidis, with N. gonorrhoeaa being the causative agent of gonorrhea and N. meningitidis being one of the causes of bacterial meningitis.[16]
Metabolism
[edit]Culture and identification
[edit]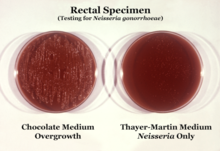

N. gonorrhoeae is usually isolated on Thayer–Martin agar (or VPN) agar in an atmosphere enriched with 3-7% carbon dioxide.[11] Thayer–Martin agar is a chocolate agar plate (heated blood agar) containing nutrients and antimicrobials (vancomycin, colistin, nystatin, and trimethoprim). This agar preparation facilitates the growth of Neisseria species while inhibiting the growth of contaminating bacteria and fungi. Martin Lewis and New York City agar are other types of selective chocolate agar commonly used for Neisseria growth.[11] N. gonorrhoeae is oxidase positive (possessing cytochrome c oxidase) and catalase positive (able to convert hydrogen peroxide to oxygen).[11] When incubated with the carbohydrates lactose, maltose, sucrose, and glucose, N. gonorrhoeae will oxidize only the glucose.[11]
Life Cycle
[edit]Surface molecules
[edit]On its surface, N. gonorrhoeae bears hair-like pili, surface proteins with various functions, and sugars called lipooligosaccharides. The pili mediate adherence, movement, and DNA exchange. The Opa proteins interact with the immune system, as do the porins. Lipooligosaccharide is an endotoxin that provokes an immune response. All are antigenic and all exhibit antigenic variation (see below). The pili exhibit the most variation. The pili, Opa proteins, porins, and even the lipooligosaccharide have mechanisms to inhibit the immune response, making asymptomatic infection possible.[17]
Type IV Pili
[edit]Dynamic polymeric protein filaments called type IV pili allow N. gonorrhoeae to do many bacterial processes including adhesion to surfaces, transformation competence, twitching motility, and immune response evasions.[18] To enter the host the bacteria uses the pili to adhere to and penetrate mucosal surfaces. The pili are a pivotal virulence factor for N. gonorrhoeae; without them, the bacterium is unable to cause infection. For motility, individual bacteria use their pili in a manner that resembles a grappling hook: first, they are extended from the cell surface and attach to a substrate. Subsequent pilus retraction drags the cell forward. The resulting movement is referred to as twitching motility. N. gonorrhoeae is able to pull 100,000 times its own weight, and the pili used to do so are amongst the strongest biological motors known to date, exerting one nanonewton. The PilF and PilT ATPase proteins are responsible for powering the extension and retraction of the type IV pilus, respectively. The adhesive functions of the gonococcal pilus play a role in microcolony aggregation and biofilm formation. These pilus are also used to avoid immune responses from the cell they are invading by having their type IV pili antigenically vary. The main pilus filament is replaced by variable DNA sequences very frequently.[18] By doing this process rapidly, they are able to create a diversity of pili on their surface and evade the host cell's immune response.
Opa Proteins
[edit]Phase-variable opacity-associated (Opa) adhesin proteins are used by N. gonorrhoeae as part of evading immune response in a host cell. At least 12 Opa proteins are known and the many variations of surface proteins make recognizing N. gonorrhoeae and mounting a defense by immune cells more difficult.[19] Opa proteins are located in the outer membrane and facilitate a response when the bacteria interacts with a variety of host cells. These proteins bind to various epithelial cells, and allow N. gonorrhoeae to increase the length of infection as well as increase the amount of invasion into other host cells.[20]
Lipooligosaccharide
[edit]Lipooligosaccharide is a low-weight version of lipopolysaccharide present on the surfaces of most other Gram-negative bacteria. It is a sugar (saccharide) side chain attached to lipid A (thus "lipo-") in the outer membrane coating the cell wall of the bacteria. The root "oligo" refers to the fact that it is a few sugars shorter than the typical lipopolysaccharide.[5] As an endotoxin, it provokes inflammation. The shedding of lipooligosaccharide by the bacteria is responsible for local injury in, for example, pelvic inflammatory disease.[5] Although its main function is as an endotoxin, lipooligosaccharide may disguise itself with host sialic acid and block initiation of the complement cascade.[5]
Antigenic variation
[edit]N. gonorrhoeae evades the immune system through a process called antigenic variation.[21] This process allows N. gonorrhoeae to recombine its genes and alter the antigenic determinants (sites where antibodies bind), such as the Type IV pili,[22] that adorn its surface.[5] Simply stated, the chemical composition of molecules is changed due to changes at the genetic level.[8] N. gonorrhoeae is able to vary the composition of its pili, and lipooligosaccharide; of these, the pili exhibit the most antigenic variation due to chromosomal rearrangement.[9][5] The PilS gene is an example of this ability to rearrange as its combination with the PilE gene is estimated to produce over 100 variants of the PilE protein.[8] These changes allow for adjustment to the differences in the local environment at the site of infection, evasion of recognition by targeted antibodies, and contribute to the lack of an effective vaccine.[8]
In addition to the ability to rearrange the genes it already has, it is also naturally competent to acquire new DNA (via plasmids), via its type IV pilus, specifically proteins PilQ and PilT.[23] These processes allow N. gonorrhoeae to acquire/spread new genes, disguise itself with different surface proteins, and prevent the development of immunological memory – an ability which has led to antibiotic resistance and has also impeded vaccine development.[24]
Phase variation
[edit]Phase variation is similar to antigenic variation, but instead of changes at the genetic level altering the composition of molecules, these genetic changes result in the activation or deactivation of a gene.[8] Phase variation most often arises from a frameshift in the expressed gene.[8] The Opacity, or Opa, proteins of N. gonorrhoeae rely strictly on phase variation.[8] Every time the bacteria replicate, they may switch multiple Opa proteins on or off through slipped-strand mispairing. That is, the bacteria introduce frameshift mutations that bring genes in or out of frame. The result is that different Opa genes are translated every time.[5] Pili are varied by antigenic variation, but also phase variation.[8] Frameshifts occur in both the pilE and pilC genes, effectively turning off the expression of pili in situations when they are not needed, such as after colonization when N. gonorrhoeae survives within cells as opposed to on their surfaces.[8]
Survival of gonococci
[edit]After gonococci invade and transcytose the host epithelial cells, they land in the submucosa, where neutrophils promptly consume them.[5] The pili and Opa proteins on the surface may interfere with phagocytosis,[9] but most gonococci end up in neutrophils. The exudates from infected individuals contain many neutrophils with ingested gonococci. Neutrophils release an oxidative burst of reactive oxygen species in their phagosomes to kill the gonococci.[25] However, a significant fraction of the gonococci can resist killing through the action of their catalase[5] which breaks down reactive oxygen species and is able to reproduce within the neutrophil phagosomes.[26]
Stohl and Seifert showed that the bacterial RecA protein, which mediates repair of DNA damage, plays an important role in gonococcal survival.[27] Michod et al. have suggested that N. gonorrhoeae may replace DNA damaged in neutrophil phagosomes with DNA from neighboring gonococci.[28] The process in which recipient gonococci integrate DNA from neighboring gonococci into their genome is called transformation.[29]

Genome
[edit]The genomes of several strains of N. gonorrhoeae have been sequenced. Most of them are about 2.1 Mb in size and encode 2,100 to 2,600 proteins (although most seem to be in the lower range).[30] For instance, strain NCCP11945 consists of one circular chromosome (2,232,025 bp) encoding 2,662 predicted open reading frames (ORFs) and one plasmid (4,153 bp) encoding 12 predicted ORFs. The estimated coding density over the entire genome is 87%, and the average G+C content is 52.4%, values that are similar to those of strain FA1090. The NCCP11945 genome encodes 54 tRNAs and four copies of 16S-23S-5S rRNA operons.[31]
Horizontal gene transfer
[edit]Horizontal gene transfer, also termed lateral gene transfer, is the sharing of genetic information amongst living organisms.[32] This transmission of information is a driving force of antibiotic resistance in N. gonorrhoeae.[33] [34] Studies have identified that N. gonorrhoeae has obtained methods of antimicrobial resistance by way of horizontal gene transfer from other Neisseria species including N. lactamica, N. macacae, and N. mucosa. [35]
N. gonorrhoeae is naturally competent, meaning that it has the ability to acquire DNA from its environment through transformation and incorporate the sequences into its own genome. [34] Transformation in N. gonorrhoeae is performed by the type IV pilus, where the DNA is bound and brought into the cell, followed by processing and homologous recombination.[36]
In 2011, researchers at Northwestern University found evidence of a human DNA fragment in a N. gonorrhoeae genome, the first example of horizontal gene transfer from humans to a bacterial pathogen.[37][38]
Disease
[edit]Symptoms
[edit]Symptoms of infection with N. gonorrhoeae differ depending on the site of infection and many infections are asymptomatic independent of sex.[39][17][6] It is important to note that depending on the route of transmission, N. gonorrhoeae may cause infection of the throat (pharyngitis) or infection of the anus/rectum (proctitis).[40][9]
Disseminated gonococcal infections can occur when N. gonorrhoeae enters the bloodstream, often spreading to the joints and causing a rash (dermatitis-arthritis syndrome).[40] Dermatitis-arthritis syndrome results in joint pain (arthritis), tendon inflammation (tenosynovitis), and painless non-pruritic (non-itchy) dermatitis.[9] Disseminated infection and pelvic inflammatory disease in women tend to begin after menses due to reflux during menses, facilitating spread.[40] In rare cases, disseminated infection may cause infection of the meninges of the brain and spinal cord (meningitis) or infection of the heart valves (endocarditis).[40][41]
Male
[edit]In symptomatic men, the primary symptom of genitourinary infection is urethritis – burning with urination (dysuria), increased urge to urinate, and a pus-like (purulent) discharge from the penis. The discharge may be foul smelling.[40] If untreated, scarring of the urethra may result in difficulty urinating. Infection may spread from the urethra in the penis to nearby structures, including the testicles (epididymitis/orchitis), or to the prostate (prostatitis).[40][9][42] Men who have had a gonorrhea infection have a significantly increased risk of having prostate cancer.[43]
Female
[edit]
In symptomatic women, the primary symptoms of genitourinary infection are increased vaginal discharge, burning with urination (dysuria), increased urge to urinate, pain with intercourse, or menstrual abnormalities. Pelvic inflammatory disease results if N. gonorrhoeae ascends into the pelvic peritoneum (via the cervix, endometrium, and fallopian tubes). The resulting inflammation and scarring of the fallopian tubes can lead to infertility and increased risk of ectopic pregnancy.[40] Pelvic inflammatory disease develops in 10 to 20% of the females infected with N. gonorrhoeae.[40]
Neonates (perinatal infection)
[edit]In perinatal infection, the primary manifestation is infection of the eye (neonatal conjunctivitis or ophthalmia neonatorum) when the newborn is exposed to N. gonorrhoeae in the birth canal. The eye infection can lead to corneal scarring or perforation, ultimately resulting in blindness. If the newborn is exposed during birth, conjunctivitis occurs within 2–5 days after birth and is severe.[40][41] Gonococcal ophthalmia neonatorum, once common in newborns, is prevented by the application of erythromycin (antibiotic) gel to the eyes of babies at birth as a public health measure. Silver nitrate is no longer used in the United States.[41][40]
Transmission
[edit]N. gonorrhoeae is transmitted through vaginal, oral, or anal sex; nonsexual transmission is unlikely in adult infection.[6] It can also be transmitted to the newborn during passage through the birth canal if the mother has untreated genitourinary infection. Given the high rate of asymptomatic infection, all pregnant women should be tested for gonorrhea infection.[6] However, communal baths, towels or fabric, rectal thermometers and caregivers hands have been implicated as means of transmission in the pediatric setting.[44] Kissing has also been implicated as a theoretical means of transmission in the gay male population, based on newer studies.[45][46][47]
Traditionally, the bacterium was thought to move attached to spermatozoa, but this hypothesis did not explain female to male transmission of the disease. A recent study suggests that rather than "surf" on wiggling sperm, N. gonorrhoeae bacteria use pili to anchor onto proteins in the sperm and move through coital liquid.[48]
Infection
[edit]For N. gonorrhoeae, the first step after successful transmission is adherence to the epithelial cells found at the mucosal site that is infected.[49] The bacterium relies on type IV pili that attach and retract, pulling N. gonorrhoeae toward the epithelial membrane where its surface proteins, such as opacity proteins, can interact directly.[49] After adherence, N. gonorrhoeae replicates itself and forms microcolonies.[50] While colonizing, N. gonorrhoeae has the potential to transcytose across the epithelial barrier and work its way in to the bloodstream.[14] During growth and colonization, N. gonorrhoeae stimulates the release of cytokines and chemokines from host immune cells that are pro-inflammatory.[14] These pro-inflammatory molecules result in the recruitment of macrophages and neutrophils.[8] These phagocytic cells typically take in foreign pathogens and destroy them, but N. gonorrhoeae has evolved many mechanisms that allow it to survive within these immune cells and thwart the attempts at elimination.[8]
Prevention
[edit]Transmission is reduced by using latex barriers (e.g. condoms or dental dams) during sex and by limiting sexual partners.[7] Condoms and dental dams should be used during oral and anal sex, as well. Spermicides, vaginal foams, and douches are not effective for prevention of transmission.[5]
Vaccine Development
[edit]Treatment
[edit]The current treatment recommended by the CDC is an injected single dose of ceftriaxone (a third-generation cephalosporin).[51] Sexual partners (defined by the CDC as sexual contact within the past 60 days)[12] should also be notified, tested, and treated.[7][51] It is important that if symptoms persist after receiving treatment of N. gonorrhoeae infection, a reevaluation should be pursued.[51]
Laboratory Tests
[edit]Antibiotic resistance
[edit]Antibiotic resistance in gonorrhea has been noted beginning in the 1940s. Gonorrhea was treated with penicillin, but doses had to be progressively increased to remain effective. By the 1970s, penicillin- and tetracycline-resistant gonorrhea emerged in the Pacific Basin. These resistant strains then spread to Hawaii, California, the rest of the United States, Australia and Europe. Fluoroquinolones were the next line of defense, but soon resistance to this antibiotic emerged, as well. Since 2007, standard treatment has been third-generation cephalosporins, such as ceftriaxone, which are considered to be our "last line of defense".[52][53] Recently, a high-level ceftriaxone-resistant strain of gonorrhea called H041 was discovered in Japan. Lab tests found it to be resistant to high concentrations of ceftriaxone, as well as most of the other antibiotics tested. Within N. gonorrhoeae, genes exist that confer resistance to every single antibiotic used to cure gonorrhea, but thus far they do not coexist within a single gonococcus. However, because of N. gonorrhoeae's high affinity for horizontal gene transfer, antibiotic-resistant gonorrhea is seen as an emerging public health threat.[53]
Prior to 2007, fluoroquinolones were a common treatment recommendation for gonorrhoeae. The CDC stopped suggesting these systemic bacterial agents once a resistant strain of N. gonorrhoeae emerged in the United States. The removal of fluoroquinolones as a potential treatment left cephalosporins as the only viable antimicrobial option for gonorrhea treatment. Wary of further gonococcal resistance, the CDC's recommendations shifted in 2010 to a dual therapy strategy--cephalosporin with either azithromycin or doxycycline. Despite these efforts, resistant N. gonorrhoeae had been reported in five continents by 2011, further limiting treatment options and recommendations. Antimicrobial resistance is not universal and N. gonorrhoeae strains in the United States continue to respond to a combination regimen of ceftriaxone and azithromycin.[54]
Serum resistance
[edit]As a Gram negative bacteria, N. gonorrhoeae requires defense mechanisms to protect itself against the complement system (or complement cascade), whose components are found with human serum.[17] There are three different pathways that activate this system however, they all result in the activation of complement protein 3 (C3).[55] A cleaved portion of this protein, C3b, is deposited on pathogenic surfaces and results in opsonization as well as the downstream activation of the membrane attack complex.[55] N. gonorrhoeae has several mechanisms to avoid this action.[14] As a whole, these mechanisms are referred to as serum resistance.[14]
History
[edit]Name origin
[edit]Neisseria gonorrhoeae is named for Albert Neisser, who isolated it as the causative agent of the disease gonorrhea in 1878.[14][3] Galen (130 AD) coined the term "gonorrhea" from the Greek gonos which means "seed" and rhoe which means "flow".[56][8] Thus, gonorrhea means "flow of seed", a description referring to the white penile discharge, assumed to be semen, seen in male infection.[14]
Discovery
[edit]In 1878, Albert Neisser isolated and visualized N. gonorrhoeae diplococci in samples of pus from 35 men and women with the classic symptoms of genitourinary infection with gonorrhea – two of whom also had infections of the eyes.[8] In 1882, Leistikow and Loeffler were able to grow the organism in culture.[14] Then in 1883, Max Bockhart proved conclusively that the bacterium isolated by Albert Neisser was the causative agent of the disease known as gonorrhea by inoculating the penis of a healthy man with the bacteria.[8] The man developed the classic symptoms of gonorrhea days after, satisfying the last of Koch's postulates. Until this point, researchers debated whether syphilis and gonorrhea were manifestations of the same disease or two distinct entities.[57][8] One such 18th-century researcher, John Hunter, tried to settle the debate in 1767[8] by inoculating a man with pus taken from a patient with gonorrhea. He erroneously concluded that both syphilis and gonorrhea were indeed the same disease when the man developed the copper-colored rash that is classic for syphilis.[55][57] Although many sources repeat that Hunter inoculated himself,[55][14] others have argued that it was in fact another man.[58] After Hunter's experiment other scientists sought to disprove his conclusions by inoculating other male physicians, medical students,[14] and incarcerated men with gonorrheal pus, who all developed the burning and discharge of gonorrhea. One researcher, Ricord, took the initiative to perform 667 inoculations of gonorrheal pus on patients of a mental hospital, with zero cases of syphilis.[8][14] Notably, the advent of penicillin in the 1940s made effective treatments for gonorrhea available.[citation needed]
See also
[edit]References
[edit]- ^ Euzéby JP, Parte AC. "Genus Neisseria". List of Prokaryotic Names with Standing in Nomenclature (LPSN). Retrieved 7 July 2017.
- ^ Neisser A (1879). "Ueber eine der Gonorrhoe eigentümliche Micrococusform" [About a micrococus form peculiar to gonorrhea]. Centralblatt für die medizinischen Wissenschaften (in German). 17 (28): 497–500.
- ^ a b c O'Donnell JA, Gelone SP (2009). "Bacterial Cause of PID: Gonorrhoeae". Pelvic Inflammatory Disease. Infobase Publishing. ISBN 978-1-4381-0159-0.
- ^ Mahapure K, Singh A (August 2023). "A Review of Recent Advances in Our Understanding of Neisseria gonorrhoeae". Cureus. 15 (8): e43464. doi:10.7759/cureus.43464. PMC 10498933. PMID 37711920.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b c d e f g h i j k Ryan KJ, Ray CG, eds. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. ISBN 978-0-8385-8529-0.[page needed]
- ^ a b c d "Detailed STD Facts - Gonorrhea". www.cdc.gov. 26 September 2017. Retrieved 7 December 2017.
- ^ a b c d e "2015 Sexually Transmitted Diseases Treatment Guidelines". CDC. Centers for Disease Control and Prevention, U.S. Department of Health & Human Services. Archived from the original on 22 December 2015.
- ^ a b c d e f g h i j k l m n o p q Hill SA, Masters TL, Wachter J (September 2016). "Gonorrhea - an evolving disease of the new millennium". Microbial Cell. 3 (9): 371–389. doi:10.15698/mic2016.09.524 (inactive 13 September 2024). PMC 5354566. PMID 28357376.
{{cite journal}}: CS1 maint: DOI inactive as of September 2024 (link) - ^ a b c d e f g Levinson W (2014). Review of medical microbiology and immunology. McGraw-Hill Education. ISBN 978-0-07-181811-7. OCLC 871305336.[page needed]
- ^ "Final Recommendation Statement: Chlamydia and Gonorrhea: Screening". U.S. Preventive Services Task Force. Retrieved 7 December 2017.
- ^ a b c d e Ng LK, Martin IE (January 2005). "The laboratory diagnosis of Neisseria gonorrhoeae". The Canadian Journal of Infectious Diseases & Medical Microbiology. 16 (1): 15–25. doi:10.1155/2005/323082. PMC 2095009. PMID 18159523.
- ^ a b "Gonococcal Infections - 2015 STD Treatment Guidelines". 4 January 2018.
- ^ MacKenzie CR, Henrich B, eds. (2012). Diagnosis of sexually transmitted diseases: methods and protocols. Humana Press. ISBN 978-1-61779-936-5. OCLC 781681739.
- ^ a b c d e f g h i j k Quillin SJ, Seifert HS (April 2018). "Neisseria gonorrhoeae host adaptation and pathogenesis". Nature Reviews. Microbiology. 16 (4): 226–240. doi:10.1038/nrmicro.2017.169. PMC 6329377. PMID 29430011.
- ^ Costa-Lourenço AP, Barros Dos Santos KT, Moreira BM, Fracalanzza SE, Bonelli RR (1 October 2017). "Antimicrobial resistance in Neisseria gonorrhoeae: history, molecular mechanisms and epidemiological aspects of an emerging global threat". Brazilian Journal of Microbiology. 48 (4): 617–628. doi:10.1016/j.bjm.2017.06.001. PMC 5628311. PMID 28754299.
- ^ Ladhani SN, Lucidarme J, Parikh SR, Campbell H, Borrow R, Ramsay ME (June 2020). "Meningococcal disease and sexual transmission: urogenital and anorectal infections and invasive disease due to Neisseria meningitidis". Lancet. 395 (10240): 1865–1877. doi:10.1016/S0140-6736(20)30913-2. PMID 32534649. S2CID 219701418.
- ^ a b c Edwards JL, Apicella MA (October 2004). "The molecular mechanisms used by Neisseria gonorrhoeae to initiate infection differ between men and women". Clinical Microbiology Reviews. 17 (4): 965–81, table of contents. doi:10.1128/CMR.17.4.965-981.2004. PMC 523569. PMID 15489357.
- ^ a b Green LR, Cole J, Parga EF, Shaw JG (1 January 2022). Kelly RK, Poole DJ (eds.). "Neisseria gonorrhoeae physiology and pathogenesis". Advances in Microbial Physiology. 80. Academic Press: 35–83. doi:10.1016/bs.ampbs.2022.01.002. ISBN 978-0-323-98869-8. PMID 35489793.
- ^ STI Awareness: Gonorrhea. Planned Parenthood Advocates of Arizona. 11 April 2011. Retrieved 31 August 2011.
- ^ Sadarangani M, Pollard AJ, Gray-Owen SD (May 2011). "Opa proteins and CEACAMs: pathways of immune engagement for pathogenic Neisseria". FEMS Microbiology Reviews. 35 (3): 498–514. doi:10.1111/j.1574-6976.2010.00260.x. PMID 21204865.
- ^ Stern A, Brown M, Nickel P, Meyer TF (October 1986). "Opacity genes in Neisseria gonorrhoeae: control of phase and antigenic variation". Cell. 47 (1): 61–71. doi:10.1016/0092-8674(86)90366-1. PMID 3093085. S2CID 21366517.
- ^ Cahoon LA, Seifert HS (September 2011). "Focusing homologous recombination: pilin antigenic variation in the pathogenic Neisseria". Molecular Microbiology. 81 (5): 1136–1143. doi:10.1111/j.1365-2958.2011.07773.x. PMC 3181079. PMID 21812841.
- ^ Obergfell KP, Seifert HS (February 2015). "Mobile DNA in the pathogenic Neisseria". Microbiology Spectrum. 3 (3). doi:10.1128/microbiolspec.MDNA3-0015-2014. PMC 4389775. PMID 25866700.
- ^ Aas FE, Wolfgang M, Frye S, Dunham S, Løvold C, Koomey M (November 2002). "Competence for natural transformation in Neisseria gonorrhoeae: components of DNA binding and uptake linked to type IV pilus expression". Molecular Microbiology. 46 (3): 749–760. doi:10.1046/j.1365-2958.2002.03193.x. PMID 12410832. S2CID 21854666.
- ^ Simons MP, Nauseef WM, Apicella MA (April 2005). "Interactions of Neisseria gonorrhoeae with adherent polymorphonuclear leukocytes". Infection and Immunity. 73 (4): 1971–1977. doi:10.1128/iai.73.4.1971-1977.2005. PMC 1087443. PMID 15784537.
- ^ Escobar A, Rodas PI, Acuña-Castillo C (2018). "Macrophage-Neisseria gonorrhoeae Interactions: A Better Understanding of Pathogen Mechanisms of Immunomodulation". Frontiers in Immunology. 9: 3044. doi:10.3389/fimmu.2018.03044. PMC 6309159. PMID 30627130.
- ^ Stohl EA, Seifert HS (November 2006). "Neisseria gonorrhoeae DNA recombination and repair enzymes protect against oxidative damage caused by hydrogen peroxide". Journal of Bacteriology. 188 (21): 7645–7651. doi:10.1128/JB.00801-06. PMC 1636252. PMID 16936020.
- ^ Michod RE, Bernstein H, Nedelcu AM (May 2008). "Adaptive value of sex in microbial pathogens". Infection, Genetics and Evolution. 8 (3): 267–285. Bibcode:2008InfGE...8..267M. doi:10.1016/j.meegid.2008.01.002. PMID 18295550.
- ^ Blokesch M (November 2016). "Natural competence for transformation". Current Biology. 26 (21): R1126–R1130. Bibcode:2016CBio...26R1126B. doi:10.1016/j.cub.2016.08.058. PMID 27825443.
- ^ "Neisseria gonorrhoeae genome statistics". Broad Institute. Retrieved 8 April 2017.
- ^ Chung GT, Yoo JS, Oh HB, Lee YS, Cha SH, Kim SJ, et al. (September 2008). "Complete genome sequence of Neisseria gonorrhoeae NCCP11945". Journal of Bacteriology. 190 (17): 6035–6036. doi:10.1128/JB.00566-08. PMC 2519540. PMID 18586945.
- ^ Burmeister AR (July 2015). "Horizontal Gene Transfer". Evolution, Medicine, and Public Health. 2015 (1): 193–194. doi:10.1093/emph/eov018. PMC 4536854. PMID 26224621.
- ^ Unemo M, Shafer WM (August 2011). "Antibiotic resistance in Neisseria gonorrhoeae: origin, evolution, and lessons learned for the future". Annals of the New York Academy of Sciences. 1230 (1): E19–E28. Bibcode:2011NYASA1230E..19U. doi:10.1111/j.1749-6632.2011.06215.x. PMC 4510988. PMID 22239555.
- ^ a b Spencer-Smith R, Roberts S, Gurung N, Snyder LA (August 2016). "DNA uptake sequences in Neisseria gonorrhoeae as intrinsic transcriptional terminators and markers of horizontal gene transfer". Microbial Genomics. 2 (8): e000069. doi:10.1099/mgen.0.000069. PMC 5320588. PMID 28348864.
- ^ Manoharan-Basil SS, González N, Laumen JG, Kenyon C (17 March 2022). "Horizontal Gene Transfer of Fluoroquinolone Resistance-Conferring Genes From Commensal Neisseria to Neisseria gonorrhoeae: A Global Phylogenetic Analysis of 20,047 Isolates". Frontiers in Microbiology. 13: 793612. doi:10.3389/fmicb.2022.793612. PMC 8973304. PMID 35369513.
- ^ Hamilton HL, Dillard JP (January 2006). "Natural transformation of Neisseria gonorrhoeae: from DNA donation to homologous recombination". Molecular Microbiology. 59 (2): 376–385. doi:10.1111/j.1365-2958.2005.04964.x. PMID 16390436.
- ^ Anderson MT, Seifert HS (May 2011). "Neisseria gonorrhoeae and humans perform an evolutionary LINE dance". Mobile Genetic Elements. 1 (1): 85–87. doi:10.4161/mge.1.1.15868. PMC 3190277. PMID 22016852.
- ^ Anderson MT, Seifert HS (2011). "Opportunity and means: horizontal gene transfer from the human host to a bacterial pathogen". mBio. 2 (1): e00005–e00011. doi:10.1128/mBio.00005-11. PMC 3042738. PMID 21325040.
- ^ Detels R, Green AM, Klausner JD, Katzenstein D, Gaydos C, Handsfield H, et al. (June 2011). "The incidence and correlates of symptomatic and asymptomatic Chlamydia trachomatis and Neisseria gonorrhoeae infections in selected populations in five countries". Sexually Transmitted Diseases. 38 (6): 503–509. doi:10.1097/OLQ.0b013e318206c288. PMC 3408314. PMID 22256336.
- ^ a b c d e f g h i j Ryan KJ, Ray CG, eds. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. ISBN 978-0-8385-8529-0.[page needed]
- ^ a b c "Gonococcal Infections - 2015 STD Treatment Guidelines". www.cdc.gov. Retrieved 7 December 2017.
- ^ "Gonorrhea (the clap) Symptoms". std-gov.org. 2 April 2015.
- ^ Caini S, Gandini S, Dudas M, Bremer V, Severi E, Gherasim A (August 2014). "Sexually transmitted infections and prostate cancer risk: a systematic review and meta-analysis". Cancer Epidemiology. 38 (4): 329–338. doi:10.1016/j.canep.2014.06.002. PMID 24986642.
- ^ Goodyear-Smith F (November 2007). "What is the evidence for non-sexual transmission of gonorrhoea in children after the neonatal period? A systematic review". Journal of Forensic and Legal Medicine. 14 (8): 489–502. doi:10.1016/j.jflm.2007.04.001. PMID 17961874.
- ^ Bever L (10 May 2019). "You May Not Need to Have Sex to Transmit Gonorrhea, New Study Suggests". ScienceAlert. The Washington Post.
- ^ Chow EP, Cornelisse VJ, Williamson DA, Priest D, Hocking JS, Bradshaw CS, et al. (November 2019). "Kissing may be an important and neglected risk factor for oropharyngeal gonorrhoea: a cross-sectional study in men who have sex with men". Sexually Transmitted Infections. 95 (7): 516–521. doi:10.1136/sextrans-2018-053896. PMID 31073095. S2CID 149446915.
- ^ Tran J, Ong JJ, Bradshaw CS, Chen MY, Kong FY, Hocking JS, et al. (September 2022). "Kissing, fellatio, and analingus as risk factors for oropharyngeal gonorrhoea in men who have sex with men: A cross-sectional study". eClinicalMedicine. 51: 101557. doi:10.1016/j.eclinm.2022.101557. PMC 9294476. PMID 35865738.
- ^ Anderson MT, Dewenter L, Maier B, Seifert HS (March 2014). "Seminal plasma initiates a Neisseria gonorrhoeae transmission state". mBio. 5 (2): e01004–e01013. doi:10.1128/mBio.01004-13. PMC 3958800. PMID 24595372.
- ^ a b Pearce WA, Buchanan TM (April 1978). "Attachment role of gonococcal pili. Optimum conditions and quantitation of adherence of isolated pili to human cells in vitro". The Journal of Clinical Investigation. 61 (4): 931–943. doi:10.1172/JCI109018. PMC 372611. PMID 96134.
- ^ Higashi DL, Lee SW, Snyder A, Weyand NJ, Bakke A, So M (October 2007). "Dynamics of Neisseria gonorrhoeae attachment: microcolony development, cortical plaque formation, and cytoprotection". Infection and Immunity. 75 (10): 4743–4753. doi:10.1128/IAI.00687-07. PMC 2044525. PMID 17682045.
- ^ a b c "CDC - Gonorrhea Treatment". www.cdc.gov. 22 July 2021. Retrieved 11 April 2022.
- ^ "UK doctors advised gonorrhoea has turned drug resistant". BBC News. 10 October 2011.
- ^ a b STI Awareness: Antibiotic-Resistant Gonorrhea. Planned Parenthood Advocates of Arizona. 6 March 2012. Retrieved 6 March 2012.
- ^ Tuddenham S, Hamill MM, Ghanem KG (January 2022). "Diagnosis and Treatment of Sexually Transmitted Infections: A Review". JAMA. 327 (2): 161–172. doi:10.1001/jama.2021.23487. PMID 35015033.
- ^ a b c d Janeway Jr CA, Travers P, Walport M, Shlomchik MJ (2001). "The complement system and innate immunity". Immunobiology: The Immune System in Health and Disease. 5th Edition.
- ^ "gonorrhea | Origin and meaning of gonorrhea by Online Etymology Dictionary". www.etymonline.com. Retrieved 5 December 2017.
- ^ a b Singal A, Grover C (2016). Comprehensive Approach to Infections in Dermatology. JP Medical. p. 470. ISBN 978-93-5152-748-0.
- ^ Gladstein J (July 2005). "Hunter's chancre: did the surgeon give himself syphilis?". Clinical Infectious Diseases. 41 (1): 128, author reply 128-128, author reply 129. doi:10.1086/430834. PMID 15937780.
External links
[edit]- Todar K. "Pathogenic Neisseriae: Gonorrhea, Neonatal Ophthalmia and Meningococcal Meningitis". Todar's Online Textbook of Bacteriology.
- Gonorrhea at eMedicine
- "Neisseria gonorrhoeae". NCBI Taxonomy Browser. 485.
